HIV drug resistance: brief report 2024
This brief report summarizes recent information on HIV drug resistance (HIVDR) in the era of integrase-strand transfer inhibitors (INSTI) for HIV prevention and treatment. In this report, WHO documents
This brief report summarizes recent information on HIV drug resistance (HIVDR) in the era of integrase-strand transfer inhibitors (INSTI) for HIV prevention and treatment. In this report, WHO documents
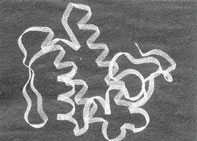
British scientists have evolved a way to make enzymes more tenacious and alter their activity

Viagra may also help women
Viruses that kill bacteria
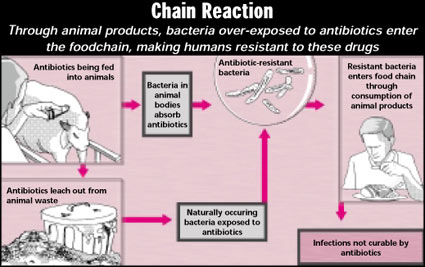
Antibiotics in meat make disease causing bacteria resistant to cures
A REPORT written by leading pharmacologists claims that about 20 per cent of the drugs prescribed in Germany offer no real clinical benefits. The report says that distinguishing between effec-tive
the drug proscar has been shown to reduce the need for surgery to treat enlarged prostrate glands. In a new study, the men who took the drug required prostrate-reduction surgery only half as often

Taxol, an effective anti cancer drug that was approved for use in the US last year, will soon be available in India at half the cost
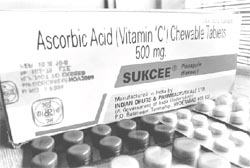
Consuming vitamin and mineral supplements can be unsafe
One of the most widely dissemina ted strains of an antibiotic-resistant bacterium responsible for hundreds of infections in European hospitals can be traced back to the 1950s, according to
Edible vaccines made from plants? This seemingly unusual proposition is on the verge of becoming a reality. Preliminary studies in the US indicate that genetically altered potatoes, bananas, alfalfa